This piece was written by Dr Tasmin Symons and Professor Peter Gething from the Malaria Atlas Project research team, who led the analysis and interpretation of the findings.
Summary
This analysis demonstrates that malaria control interventions have prevented an estimated 1.57 billion cases and 5.7 million deaths in Africa since 2000, with ITNs accounting for the majority of impact. Despite this success, progress has stalled since 2015 not due to tool limitations, but primarily because intervention coverage has plateaued.
Key insights include:
- Next-generation ITNs have successfully offset losses from insecticide resistance, averting an additional 39.3 million cases between 2019 and 2024
- 2024 saw record annual impact with 134 million cases averted, demonstrating that maintaining coverage continues to prevent substantial disease
- Achieving 80% ITN coverage could restore the 2005-2015 rate of decline
- Intervention impacts vary geographically, underscoring the need for locally tailored strategies
These findings provide a foundation for strategic decisions about deploying limited resources for maximum impact as the malaria control community navigates uncertain funding and evolving biological threats.
Purpose
After 25 years of intensive malaria control efforts in Africa, understanding what has worked, where, and to what extent, remains critical for informing future policy decisions. Since the last comprehensive evaluation published in 2015 (Bhatt, S., Weiss, D., Cameron, E. et al.(2015), progress has slowed, new tools have emerged, and biological threats such as insecticide resistance have intensified. This study provides the first full accounting of malaria control impact across Africa from 2000 to 2024, quantifying reductions in transmission, clinical cases, and malaria mortality averted.
Methodology overview
A Bayesian geospatial modelling framework was used to empirically estimate how malaria control interventions impact disease burden under real-world conditions. The analysis collated 60,466 observations of Plasmodium falciparum infection prevalence (PfPR) collected across 44 countries, linking these to intervention coverage and environmental data.
Data sources included cross-sectional prevalence surveys standardised to represent infection rates in children aged 2-10 years. Intervention coverage data were derived from national malaria programmes, WHO reports, and household surveys for insecticide-treated nets (ITNs), indoor residual spraying (IRS), seasonal malaria chemoprevention (SMC), and antimalarial treatment access. Environmental covariates were obtained from satellite remote sensing, including urbanisation patterns.
The spatio-temporal model simultaneously estimated the effects of malaria control interventions while accounting for environmental factors and unexplained variation. The approach derives intervention effects directly from observational data, capturing how tools perform under routine conditions. For each intervention, counterfactual scenarios isolated the contribution of each factor. PfPR estimates were converted to clinical incidence using an established natural history model, and to mortality using country-specific untreated case fatality rates.
Key findings
Burden averted: Malaria control has prevented an estimated 1.57 billion cases (95% CI: 0.7–2.2B) and 5.7 million deaths (95% CI: 2.8–8.0M) in Africa since 2000 (Figure 1). Vector control—primarily ITNs—accounted for 71% of cases averted and 53% of deaths averted, while improved antimalarial treatment played an additional contribution in mortality reduction (Figure 1c).
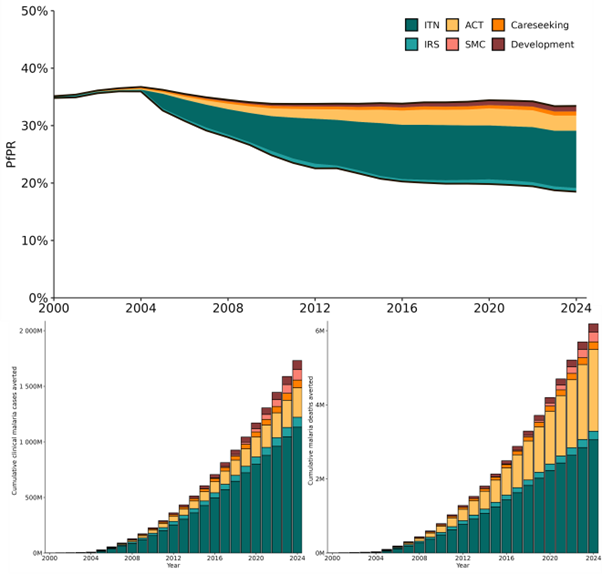
Figure 1. Estimated impact of interventions on malaria in sub-Saharan Africa 2000-2024. Estimated time-series of a. PfPR2-10 averted, b. cumulative clinical malaria cases averted and c. cumulative malaria mortality averted in malaria-endemic Africa by IRS, ITNs, ACTs, improved healthcare access and urbanisation.
Geographic variation: ITNs showed the largest relative impact in western and central Africa, where both baseline transmission and coverage were highest (Figure 2a). IRS contributed locally where high coverage was maintained, particularly in southern Africa. ACT scale-up had substantial impact across the continent, with strong effects in eastern and southern Africa (Figure 2c). Urbanisation reduced transmission notably around Lake Victoria and in expanding city peripheries (Figure 2e).
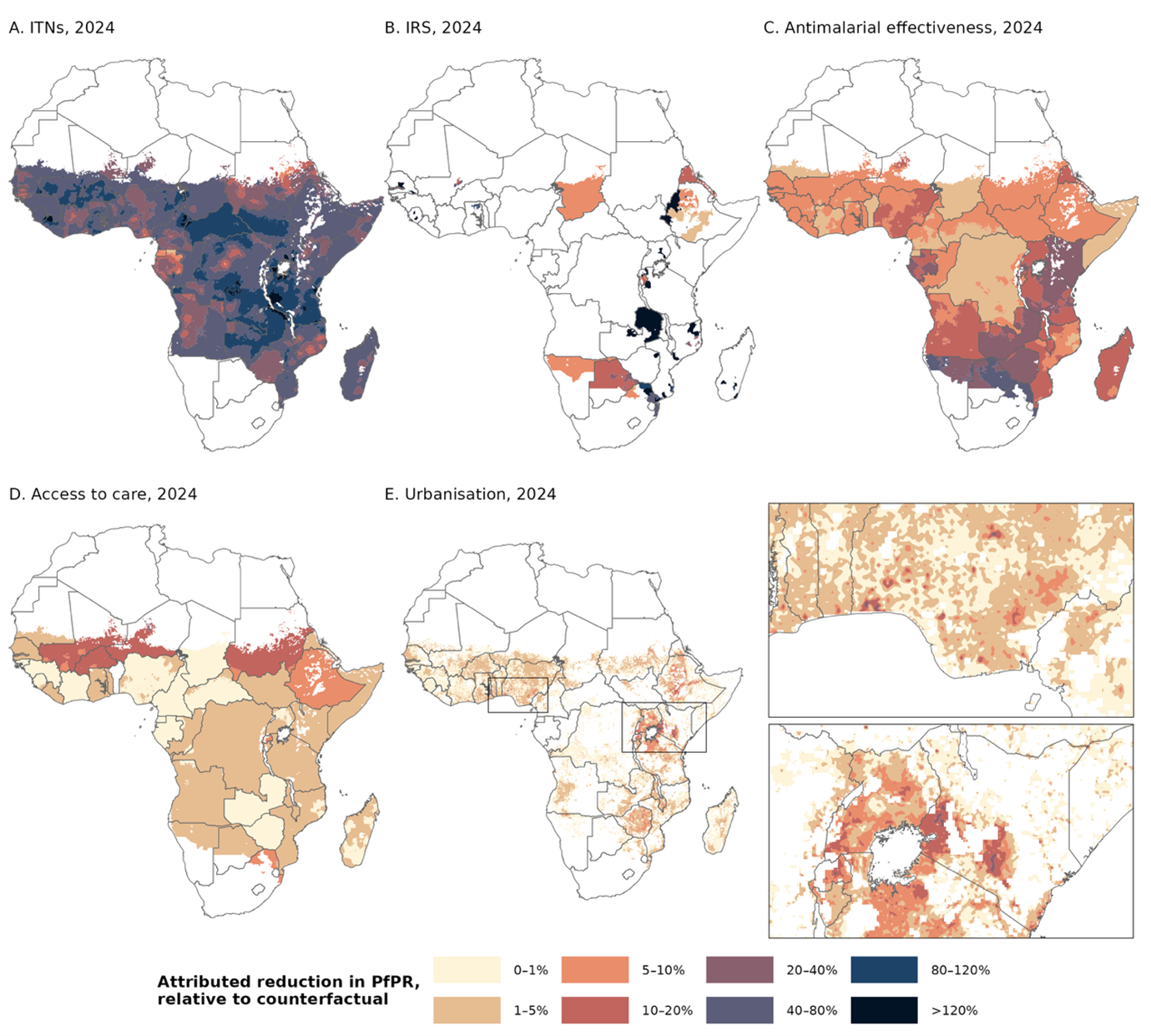
Figure 2. Contribution of each driver of transmission to reductions in PfPR. Reduction in PfPR2-10 in 2024 attributed to a. ITNs; b. IRS; c. use of ACTs versus less effective antimalarials; d. improved access to treatment with antimalarials; and e. urbanisation. The impact of each factor was calculated based on 2024 estimated coverage/levels, relative to counterfactuals where coverage/levels were either set to zero (IRS and ITNs), or held constant at 2000 levels (treatment, urbanisation). Inset shows reduction in PfPR2-10 associated with urbanisation in coastal west Africa (top) and the Lake Victoria region (bottom). Non-endemic areas and areas of no change in coverage are masked in white.
Explaining stalling progress: Average malaria infection prevalence in endemic Africa declined from 35.9% (95% CI: 34.3 –38.2%) in 2003 to 18.4% (95% CI: 17.3 –23.0%) in 2024, but most progress occurred before 2015. This analysis shows this deceleration is largely attributable to plateauing intervention coverage, rather than limitations of the tools themselves. ITN coverage has remained essentially flat since 2015, while antimalarial treatment coverage improvements slowed. The modelling suggests that achieving 80% ITN coverage across endemic areas could have resulted in prevalence maintained the 2005 –2015 rate of decline, reaching 13.7% by 2024 rather than 18.5%.
Insecticide resistance impact and mitigation: Rising pyrethroid resistance has reduced ITN effectiveness: without it, we estimate prevalence would be 17.3% rather than 18.5% in 2024. However, next-generation ITNs (containing PBO or dual active ingredients) have already averted an estimated additional 39.3 million cases between 2019 and 2024, substantially offsetting resistance-related losses (Figure 3).
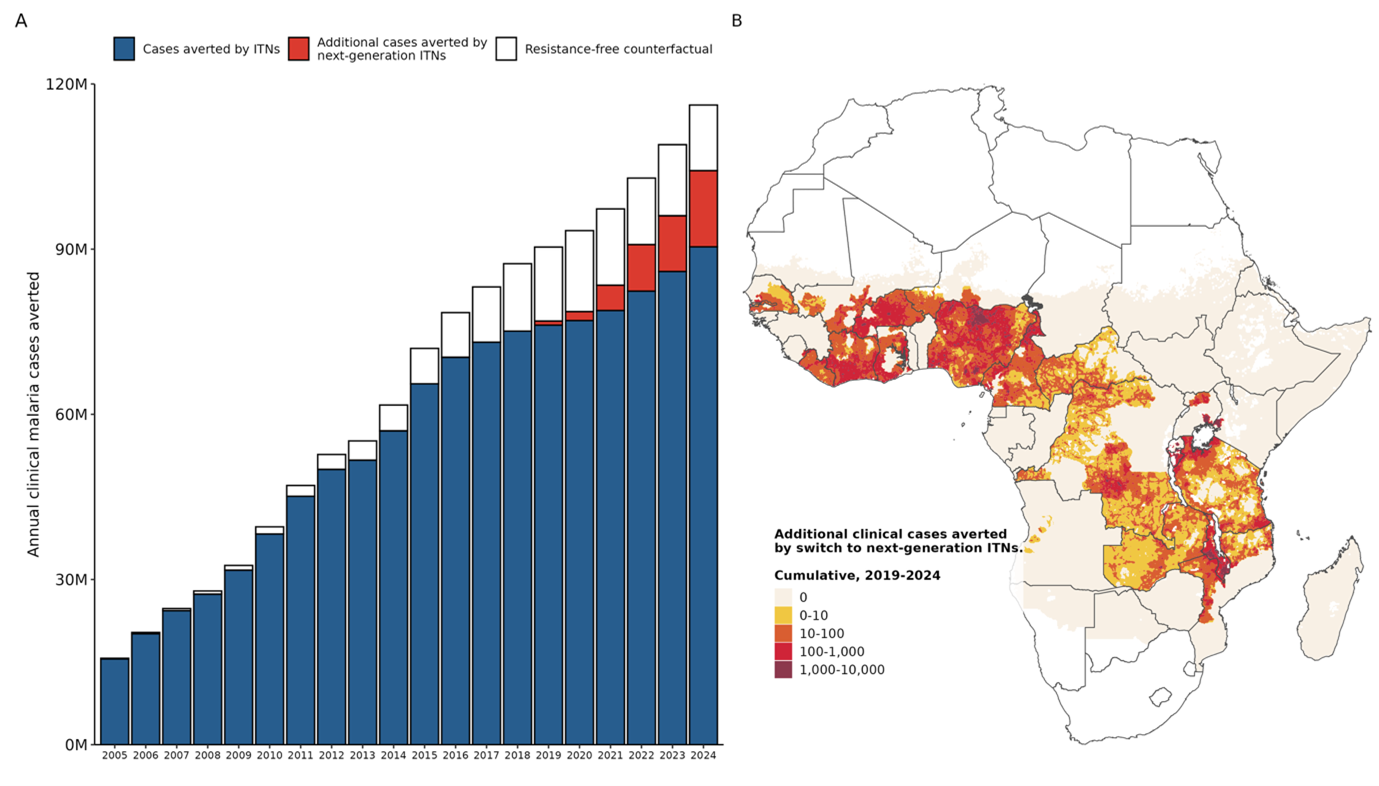
Figure 3. Impact of ITNs on clinical malaria burden in Africa. a. Estimated annual cases averted by ITNs (blue and red bars). Area coloured red indicates cases averted by next-generation ITNs, relative to a counterfactual scenario in which they did not replace conventional ITNs. The remaining area within the bar is the estimated number of additional cases which would have been averted if insecticide resistance had not developed. b. Spatial distribution of cumulative cases averted by switch to next-generation ITNs 2019-2024 (this corresponds in total to the red bars on panel a. Non-endemic areas shown in white.
Sustained ongoing impact: Despite flat disease trends, malaria control continues to avert enormous burden each year as interventions reach growing populations. More cases were averted in 2024 (134 million ; 95% CI: 93 –180M) than in any previous year, demonstrating that maintaining coverage prevents considerable disease even when further progress has stalled.
Implications for vector control
These findings carry important implications for policy and investment decisions. First, sustaining coverage is critical: the 134 million cases averted in 2024 represent lives saved and healthcare systems spared. Second, innovation continues to deliver real-world benefits, as next-generation ITNs demonstrably counter insecticide resistance at scale. Third, closing coverage gaps could restore momentum, with substantial scope for further impact of existing interventions. Fourth, the geographically heterogeneous impacts underscores the need to tailor strategies to local conditions.
Finally, sustained funding remains essential as current investments continue generating substantial health returns. This analysis provides evidence-based insight into which interventions have driven progress, why momentum has slowed since 2015, and where opportunities exist to regain it. As the malaria control community navigates uncertain funding and evolving biological threats, these findings offer a foundation for strategic decisions on deploying limited resources for maximum impact.
Additional methods, results and a full reference list can be found in the pre-print, hosted at https://malariaatlas.org/project-resources/impact-of-control-2000-2025/



